
Homepage - - - Tutorial Index
Membranous Glomerulonephritis
Membranous glomerulonephritis (MGN) is a disease characterized by subepithelial immune deposits, thickening, usually diffuse, of the glomerular capillary walls, and, in many cases, formation of perpendicular projections of material similar to the glomerular basement membrane (GBM) in the external part of this one (between podocyte cytoplasm and GBM): “spikes”. Because in this glomerulopathy inflammatory cells usually are not detected and that in some, or many of the cases, there is not prominent local inflammation, but trapping of immune complexes, some authors prefers to use the name membranous glomerulopathy or membranous glomerulonephropathy; nevertheless, the presence of immunoglobulins, complement, and membrane attack complex (MAC) (C5b-9) implies an inflammatory process (see below). Other used terms have been: membranous nephropathy and epimembranous, perimembranous or extramembranous nephropathy (or glomerulopathy).
MGN is more commonly a primary or idiopathic disease, but it also appears as a disease secondary to other conditions, mainly infections, neoplasms and systemic lupus erythematosus (SLE). In approximately 25% of cases the disease is secondary, being greater the percentage in children and elderly patients (Glassock RJ, Nephrol Dial Transplant 7(S):64-71, 1992 [PubMed link]). Histopathologic findings do not allow a differentiation between primary or secondary forms, nevertheless, some microscopic characteristics (hypercellularity, crescents) and immunopathologic findings (complement deposits that indicate activation of the classic pathway: C1q, C4) permit suspect secondary forms.
MGN is the commonest cause of nephrotic syndrome (NS) in Caucasian adults (focal and segmental glomeruloesclerosis being the commonest in Afro-American and in Hispanic patients (Arias LF, et al. Glomerular diseases in a Hispanic population: review of a regional renal biopsy database. Sao Paulo Med J. 2009;127(3):140-4. [PubMed link][Free full text]). This disease is responsible approximately for 21-35% of NS cases in adults and 1,5-9% in children. Many series show greater frequency of MGN in male patients, with a male:female relation of 2:1.
The presence of immunoglobulins (Igs) and complement components in capillary walls (subepithelial), and the morphologic and immunopathologic similarities between the experimental MGN and immunological glomerular diseases support the concept that MGN is an immune complexes mediated disease. The deposits come from circulating immune complexes in some cases, but in the majority they correspond to in situ formation by circulating antibodies that recognize native antigens of the glomerulus or antigens foreign to it that have been deposited there.
Membranous nephropathy most likely is a heterogeneous disease, although a common denominator may be that podocytes provide antigenic targets for in-situ formation of glomerular immune deposits.
Many years ago it has been well-known the morphologic similarities between MGN and the Heymann’s experimental nephritis. In this experimental model, rats are immunized against antigens of the renal cortex; the animals develop a disease clinical and morphologically similar to MGN.
Initial studies of this model suggested that the subepithelial deposits resulted from glomerular trapping of circulating immune complexes formed by circulating brush-border related antigens and the corresponding antibodies. This hypothesis was based on the observation that the glomerular disease was induced by fractions of membrane prepared from rat renal brush-border, not from glomerular extracts. Subsequently, the development of the model of passive HN in rats that received an injection of rabbit anti-rat brush-border antibodies led to the suggestion that subepithelial immune deposits could be formed without the intervention of circulating immune complexes. Other authors demonstrated that anti-brush-border antibodies could bind glomeruli in the absence of circulating brush-border-related antigen, which provided the proof of principle that immune complex formation occurred in situ. Definitive evidence establishing the role of in situ immune complex formation in the glomerular capillary wall required identification of the antigen. As a historical data, an autoantigenic target in the rat disease was identified by Kerjaschki and Farquhar (Kerjaschki D, Farquhar MG: Proc Natl Acad Sci U S A 79 : 5557 –5561, 1982 [PubMed link] [Free Full Text] / Kerjaschki D, Farquhar MG. J Exp Med 157 : 667 –686, 1983 [PubMed link] [Free Full text]) in the early 1980s as the podocyte membrane protein now called megalin. The polyspecific receptor megalin, a member of the LDL-receptor superfamily, is expressed with clathrin at the sole of podocyte foot processes (where immune complexes are formed) (Ronco P, Debiec H. Molecular pathomechanisms of membranous nephropathy: from Heymann nephritis to alloimmunization. J Am Soc Nephrol. 2005;16(5):1205-13. [PubMed link][Full Text link]).
In the human MGN in very few cases has been demonstrated antibodies against brush border antigens. In the Seventies, a Japanese group demonstrated location of tubular antigens in the immune deposits of patients with MGN (Naruse T et al, J Exp Med 144:1347-62, 1976 [PubMed link] [Free full text]), nevertheless, groups of other centers have not found such finding (Collins AB, et al, Nephron 27:297-301, 1981 [PubMed link]; Thorpe LW y Cavallo T, J Clin Lab Immunol 3:125-127, 1980 [PubMed link]; Whitworth JA, et al, Clin Nephrol 5:159-162, 1976 [PubMed link]). At the present the evidence suggests that the antigen-antibody complex (Ags-Acs) of the Heymann’s nephritis has not roll in human MGN. The variety of Ags-Acs associated with secondary forms of MGN suggests that in idiopathic forms of the disease the morphologic presentation is common to many Ags-Acs complexes.
Neutral endopeptidase - a podocyte antigen that can digest biologically active peptides - was identified as the target antigen of antibodies deposited in the subepithelial space of glomeruli in a subset of patients with antenatal membranous nephropathy. The mothers became immunized because they are deficient in neutral endopeptidase due to truncating mutations in the gene (Ronco P, Debiec H. New insights into the pathogenesis of membranous glomerulonephritis. Curr Opin Nephrol Hypertens. 2006;15:258-63. [PubMed link])
More recently (in a paper published in July 2009), a group carried out Western blotting of protein extracts from normal human glomeruli with serum samples from patients with membranous nephropathy, and found that a majority of patients with idiopathic membranous nephropathy have antibodies against a conformation-dependent epitope in M-type phospholipase A2 receptor (PLA2R), indicating that PLA2R is a major antigen in this disease" (Beck LH, et al. M-Type Phospholipase A2 Receptor as Target Antigen in Idiopathic Membranous Nephropathy. N Engl J Med. 2009;361(1):11-21. [Extract link]). A recent work (2010) reported co-localization of specific anti-aldose reductase (AR) and anti-manganese superoxide dismutase (SOD2) with IgG4 and C5b-9 in electron-dense podocyte immune deposits. The data support AR and SOD2 as renal antigens of human MN and suggest that oxidative stress may drive glomerular SOD2 expression (Prunotto M, et al. Autoimmunity in membranous nephropathy targets aldose reductase and SOD2. J Am Soc Nephrol. 2010;21(3):507-19. [PubMed link]).
Neutral endopeptidase and PLA2R are two antigens that, in nonpathologic conditions, localize at the podocyte membrane. However, anti-NEP antibody levels in adults with membranous nephropathy have not been found to be different from apparently healthy controls, suggesting that they are not involved in adult idiopathic MN. In this light, anti-NEP determination in patients seems without practical value. Data for anti-PLA2R specificity seem solid and their assessment may be extremely useful for clinicians, helping to differentiate between idiopathic and secondary forms of the disease (Murtas C, et al. Circulating antipodocyte antibodies in membranous nephropathy: new findings. Am J Kidney Dis. 2013;62(1):12-5. [PubMed link]).
In 2014, Tomas NM et al published that aproximately 2.5 to 5% of the patients with idiopathic membranous nephropathy whom we evaluated had autoantibodies against thrombospondin type-1 domain-containing 7A (THSD7A), which corresponds to 8 to 14% of the patients who are seronegative for anti-PLA2R1 antibodies. THSD7A was initially characterized as an endothelial protein that is expressed in the placental vasculature. The authors found that THSD7A is concentrated at the basal aspect of the podocyte, colocalizing with nephrin, and they did not find any expression in glomerular endothelial cells. (Tomas NM, Beck LH, 0Meyer-Schwesinger C, et al. Thrombospondin Type-1 Domain-Containing 7A in Idiopathic Membranous Nephropathy. N Engl J Med 2014; 371:2277-87 [Article on NEJM - link]; Alsharhan L, Beck LH Jr. Membranous Nephropathy: Core Curriculum 2021. Am J Kidney Dis. 2021 Mar;77(3):440-453. [PubMed link] [Free full text]; Sethi S. New 'Antigens' in Membranous Nephropathy. J Am Soc Nephrol. 2021 Feb;32(2):268-278. [PubMed link]).
At least 14 target antigens have been identified before 2023, accounting for 80%–90% of cases of MN: PLA2R, THSD7A, NELL1, SEMA3B, PCDH7, HTRA1, NTNG1, EXT1/EXT2, NCAM1, TGFBR3, CNTN1, FAT1, NDNF, PCSK6. Many of the forms of MN associated with these novel MN target antigens have distinctive clinical and pathologic phenotypes. In 2023, 17 additional putative antigens were described, significantly reducing our knowledge gap of targets in the disease. These include seizure-related 6 homolog-like 2 (SEZ6L2), vasorin (VASN), early endosomal antigen 1 (EEA1), macrophage-stimulating 1 (MST1), natriuretic peptide receptor 3 (NPR3), ficolin 3 (FCN3), CD206, cysteine-rich motor neuron protein (CRIM1), leucine-rich repeat transmembrane protein (FLRT3), insulin-degrading enzyme (IDE), reversion-inducing cysteine-rich protein with Kazal motifs (RECK), neuroligin 3 (NLGN3), peptidoglycan recognition protein 1 (PGLYRP1), vascular endothelial growth factor A (VEGFA), sulfatase 1 (SULF1), epidermal growth factor–containing fibulin extracellular matrix protein 2 (EFEMP2) and matrix protein Fraser syndrome 1 (FRAS1). Evidence regarding the pathogenicity, clinicopathologic associations and clinical role of antibodies against each of the aforementioned antigens varies in the literature (Bonilla M, et al. Hope or hype? Clinicians' dilemma in the era of ever-expanding antigens in membranous nephropathy. Nephrol Dial Transplant. 2023 Nov 30;38(12):2666-2669. [PubMed link]).
The Mayo Clinic consensus report on MN, published in 2023, proposes a 2-step classification of MN. The first step, when possible, is identification of the target antigen, based on a multistep algorithm and using a combination of serology, staining of the kidney biopsy tissue by immunofluorescence or immunohistochemistry, and/or mass spectrometry methodology. The second step is the search for a potential underlying disease or associated condition, which is particularly relevant when knowledge of the target antigen is available to direct it. The meeting acknowledges that the resources and equipment required to perform the proposed testing may not be generally available. However, the meeting consensus was that the time has come to adopt an antigen-based classification of MN because this approach will allow for accurate and specific MN diagnosis, with significant implications for patient management and targeted treatment (Sethi S, Beck LH Jr, Glassock RJ, et al. Mayo Clinic consensus report on membranous nephropathy: proposal for a novel classification. Kidney Int. 2023 Dec;104(6):1092-1102. [PubMed link]).
In MGN is necessary activation of the complement system for NS development, with formation of MAC (C5b-9) (Groggel GC, et al, J Clin Invest 72:1948-1957, 1983 [PubMed link] [Free full text]), which supports a cytolitic roll of the complement in this disease.
MGN and hepatitis B: The most frequent glomerulopathy in patients infected with hepatitis B virus is MGN followed by membranoproliferative GN. The antigens Core (HBcAg) and e (HBeAg) seem the most important in the pathogenesis of hepatitis B-associated MGN. In these cases the antigens, or their antibodies, are identified in the glomerular immune deposits. It is not clear what is first deposited: the Ag., the Ac. or the Ag-Ac complex previously formed (circulating). Prevalence of MGN in the infection is not known, but in children with MGN the carrier stage is detected in around 20% of cases, with higher rates in endemic countries. In adults the percentage of patients with MGN carrying hepatitis B virus is lower than in children. In GNM cases associated with this infection there are more frequently mesangial hypercellularity, endocapillary proliferation, subendothelial immune deposits, and tubuloreticular endothelial structures (electron microscopy). It is frequent that appears with hypocomplementemia. The prognosis of MGN in hepatitis B patients seem more favorable, with most frequency of remission and less probability of evolution to terminal renal damage.
MGN and hepatitis C: In this infection disease also secondary MGN can appear, although membranoproliferative GN is more frequent. In many studies have not been identified antigens of the virus, or Acs against these, in the glomerular deposits. Clinic expression can be similar to idiopathic MGN or it may appear with asymptomatic proteinuria.
Congenital Syphilis: MGN is a rare complication in congenital syphilis, but it is a well-recognized cause of NS in children with this infection. Other glomerular disease in congenital syphilis include nephritic syndrome and crescentic GN with rapidly progressive disease. We have seen cases with these types of glomerular disease and there is a dramatic improvement with the antibiotic treatment. Several studies have demonstrated the presence of antigens of Treponema pallidum in the immune glomerular deposits.
In SLE the histopathologic presentation is very variable and there is combination of morphologic changes: MGN with subendothelial deposits, endocapillary and/or mesangial proliferation, crescents, combination with characteristics of membranoproliferative GN, and other patterns. In the most recent lupus nephritis classification, pure MGN (class V) is only diagnosed if there are no other active lesions; if there is combination with active lesions it is diagnosed as combination of class V and class III or IV only if there are lesions with membranous characteristics in more than 50% of the tuft in more than 50% of glomeruli. Occasional subepithelial deposits and “spikes” formation are very frequent in class III and IV lupus nephritis. In most of these cases we find C1q glomerular deposits.
MGN and neoplasms: The neoplasms more frequently associated with MGN are lung, breast, colon, stomach and kidney carcinomas, leukemia and lymphomas (Hodgkin’s and non-Hodgkin’s), but there is information of MGN in many other cancer types. Incidence of cancer in patients with MGN is approximately 1%. The histologic and immunopathologic findings and the clinical presentation are similar to those of idiopathic forms of MGN. The association between MGN and neoplasms is supported by the clinical course, the immune response of the host to the tumor and the glomerular pathology, nevertheless, in very few cases is documented an antigen of the tumor, or its antibody, in glomerular deposits. It is possible that the immune response against the neoplasm, in a propitious genetic context, allow the development of MGN. The prognosis of the glomerulopathy depend on that of the neoplasm. If there are treatment and response of this last one, the MGN tends to disappear.
See Case 144 of our Case Series: MGN asociated to B cell small lymphocytic lymphoma.
Other diseases associated with MGN are: drugs or toxins (gold, penicillamine, mercury, captopril, etc.), other infections (parasitic, Streptococcus), other autoimmune diseases (rheumatoid arthritis, pemphigus, primary biliary cirrhosis, autoimmune enteropathy, Hashimoto’s disease, Graves’ disease, and so on), diabetes, sarcoydosis, cryoglobulinemia, and sick cell disease.
Clinical features: The most frequent presentation is proteinuria in nephrotic range, with or without the other findings of the complete NS. In a variable percentage of cases present as asymptomatic proteinuria. There is microscopic hematuria in most of patients, but macrohematuria is rare. Exceptionally it can appear with isolated hematuria. The renal function can be slightly altered at the time of the diagnosis in many cases, but renal failure is unusual at presentation. In 25-33% of the cases systemic hypertension is documented. MGN may appear to any age, with predilection by 4º and 5º decades of the life.
The clinical course of MGN is very variable, in many patients there is a favorable course; approximately 25% of patients will have partial or complete spontaneous remission, although, until 29% of them will present recurrence. Around 50% of patients will not present alteration of the renal function. In a small number of cases there will be a fast loss of renal function or death. This variable evolution makes difficult interpretation of clinical trials or treatment response. Treatment with steroids, clorambucil or other immunosuppressors has shown contradictory results; it does not exist at the present moment an universally accepted treatment.
Cases of post-transplant recurrent MGN have been informed, but, there are no large series that allow determining with precision the percentage of post-transplant recurrence. Since renal transplant receptors are susceptible to many causes of secondary MGN, an underlying or associated cause must be looked for. Histologically is not possible to differentiate between recurrent MGN and de novo MGN in a transplanted kidney; for this differentiation the histologic study in the native kidney is indispensable (pre-transplant).
Laboratory findings: Proteinuria is generally in nephrotic range and in many cases it is massive (>10 g/24h). There are, in addition, in most of cases, the other findings of the complete NS (hypoalbuminemia, hypercholesterolemia). Proteinuria usually is nonselective. Mmicrohematuria is common and macrohematuria is unusual. In many cases there is slight increase of serum creatinine and BUN, and in until 75% of cases there is reduction of the glomerular filtration rate. There is no hypocomplementemia (at least in idiopathic forms) and in many cases increased levels of MAC (C5b-C9) are documented. As was previously expressed, in a high percentage of cases circulating immune complexes are detected, although, as detection of increased levels of MAC, it has few importance for the differential diagnosis, since they are not specific.
Histopathology
The characteristic changes in MGN are in the glomerular capillary walls. The initial phase of the glomerulopathy is marked by subepithelial granular deposits: in the external part of the GBM, between this one and podocyte cytoplasm. Initially these deposits do not generate reaction of the GBM and therefore it is improbable that they are detected with light microscopy. In few cases the deposits are as large as to be seen with light microscopy with a good section and a good trichrome stain; they appear fuchsinophilic (red), sometimes homogenously spaced, and in the external part of the basement membrane (Figure 3); in order to see them thin histologic sections are needed and to observe with high power using immersion oil. In tangential sections of the GBM, with methenamine-silver stain, in some cases, a mottled aspect or very small orifices (“holes”) can be observed; them correspond to depressions originated by the subepithelial deposits (Figure 4), better observed with scanning electron microscope. This phase of the MGN evolution is called: STAGE I, the diagnosis can be missed if we do not have immunofluorescence (IF) or electron microscopy (EM); these deposits are immune and will be positive for IgG and, in most of cases, for C3, in addition, they are electron-dense. Without IF or EM it is very probable that we can not diagnose this stage, and possibly we will think in minimal change disease.
In STAGE II the histologic
findings with light microscopy allow an easier diagnosis. The glomerular architecture
is preserved and the capillary walls appear thickened with routine stains (Figure
1). The cellularity usually is not increased (if present it suggests a secondary
MGN) and the capillary lumina are ample. There is formation of material with
similar aspect to the GBM (although with different composition) that projects
perpendicularly to this one: “spikes” (Figure 2). These spikes are
originated in reaction to the deposits and go progressively surrounding them.
This material is composed of type IV collagen and noncollagenous components:
laminin, proteoglycans and vitronectin, and could be originated by mediators
produced by the podocyte or another mechanism that stimulates changes in the
GBM. In some sections are well observed the spaces or holes that produce the
immune deposits in the outer aspect of the GBM, with the center of the hole
corresponding to the immune deposit and the periphery to the GBM-like material.
In some cases these holes have an irregular form that gives a reticulated aspect
to the GBM (Figure 4). In some cases we find segmental cellular proliferation
(Figure 5), but in these cases we must consider the possibility of a secondary
MGN; in other cases there will also be segmental and focal sclerosing lesions
(Figure 6).

Figure 1. In GNM the alterations are demonstrated, mainly, in the capillary walls; here they appear thickened and with rigid aspect (green arrows). In some cases we find variable degrees of mesangial hypercellularity (blue arrows), in these cases we must consider the possibility of a secondary MGN. (H&E, X.400).

Figure 2. The characteristic finding that allows the diagnosis in most of cases are the perpendicular projections in the outer aspect of the GBM, seen with silver stain (arrows). In initial stages when spikes are not still formed may not be possible to do the diagnosis without immunofluorescence or electron microscopy. (Methenamine-silver, X1000).
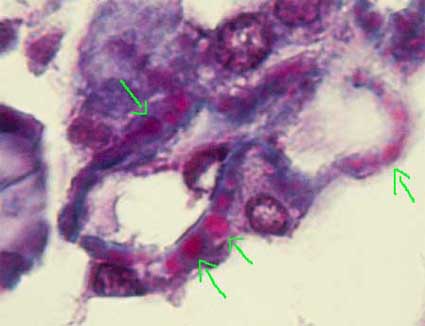
Figure 3. In cases of large subepithelial deposits is possible, in thin sections and with a good trichrome stain, to see the immune deposits with a characteristic red color (fuchsinophilic) (arrows). Even in initial stages, without “spikes”, this finding allows us to make the diagnosis of MGN. Unfortunately it is not frequent to see these deposits with light microscopy (Masson’s trichrome, X1000)..

Figure 4. In sections in which the GBM appears tangential it is possible to see spaces or holes (arrows); these holes are due to the presence of immune deposits, negative with the silver, surrounded by BGM-like (positive with the silver). Many of these holes correspond to deposits completely surrounded by the “spikes”. (Methenamine-silver, X1000).

Figure 5. With PAS stain the thickening of capillary walls is evidenced; there is no a noticeable mesangial widening. In this case there is also a segment with endocapillary proliferation (arrows); this finding must suggest the possibility of a secondary MGN. (PAS, X.400).
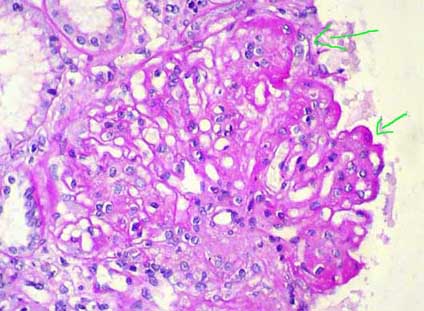
Figure 6. In some cases there are sclerosing segmental lesions (arrows). This finding not necessarily indicates a worse prognosis (PAS, X.400).
When advancing the process the material that forms the spikes increases and surround completely the deposits, forming thus new layers of GBM leaving the deposits immersed in this material. Now the deposits are seen intramembranous and with silver stain capillary walls can take an aspect in “chain” or “rosary”. This evolution point of the glomerular changes is known as STAGE III. The deposits continue being positive with the immunostaining (IF), although progressively they are less electron-dense (Figures 7 and 8).

Figure 7. When advancing the process of the disease the parietal immune deposits are progressively surrounded by GBM-like material, which gives an irregular aspect in chain or “rosary” to the GBM (arrows); these findings characterize stage III of the MGN. According to the predominant alteration in the capillary we diagnose the lesions as stage I, II, III, or IV. (Methenamine-silver, X1000).
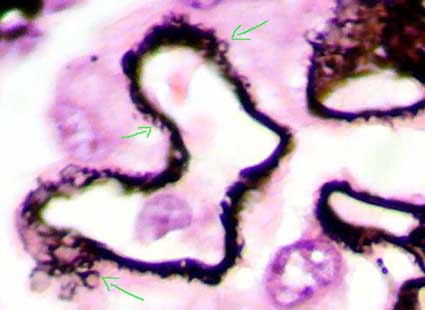
Figure 8. In this microphotography is possible demonstrate better the capillary wall lesions in stage III MGN; see as the material that appears in the external part of the GBM (black with the silver) forms circles or ring that completely surrounds the immune deposits (arrows). (Methenamine-silver, X1000).
In STAGE IV the GBM is irregularly thickened, without the presence of electron-dense deposits or holes. In this phase it is considered that the deposits have been resorpted leaving this irregular aspect. In these cases the diagnosis is sustained by the presence of other areas with lesions in stage II or III.
In many cases there is a mixed appearance, with areas presenting several stages. In order to classify these cases a good observation is required to determine the dominant pattern.
The histopathologic stages are progressive, nevertheless, although they present some correlation with the clinical evolution of the disease, there is no a perfect correlation between the stage and the prognosis. Remissions in anyone of this stages is possible, and progression to chronic renal failure has been described in stages I and II. It is not clear if these stages evolve in periods of time more or less determined. In Figure 9 appears the scheme of the different stages of MGN.

Figure 9. This scheme represents the characteristics of the different MGN stages. In stage I (blue dark arrows) the deposits have still not generated reaction in the GBM and therefore they are not accompanied by spikes. In stage II the reaction produced in the outer aspect of the GBM is seen as perpendicular projections: “spikes” that try to surround the deposits (green arrows). In stage III the GBM-like material has surrounded completely the deposits (red arrows). And in stage IV the GBM is very thickened and irregular and the deposits have disappeared almost completely (blue clear arrows). (Scheme on a microphotography of a section stained with Masson’s trichrome, X1000).
Other changes described in MGN are segmental sclerosis, lobulation of the tuft, mesangial hypercellularity, presence of inflammatory cells and necrosis, nevertheless, in these cases we must suspect a secondary form. In some works have been documented coexistence of MGN and IgA nephropathy, MGN and diabetes, and MGN and crescentic GN. Occasionally there are cases of MGN with crescents, in these cases the course is severe with poor prognosis; in several of these cases antibodies anti-GBM have been detected.
The spikes formation in GBM is “almost” diagnostic of MGN, nevertheless, I have seen some cases in which the similar aspect has generated confusion: amyloidosis with extensive formation of perpendicular projections in the GBM outer aspect, fibrillary and immunotactoid GN, and lecithin cholesterol acyl transferase deficiency.
The interstitium, tubules and vessel show unspecific changes. Frequently droplets of protein resorption or vacuolated aspect are observed in the tubular cells cytoplasm. Interstitial fibrosis and tubular atrophy correlate with the severity of the chronic damage and they are good prognosis indicators, reason why they would be quantified or semiquantified (mild - moderate - severe). The causes of tubulointerstitial damage, like in many glomerulopatías, seem to be related to alteration of the glomerular circulation and secondary atrophy. Proteinuria also can play an important role in the tubular damage.
Immunofluorescence
The characteristic immunopathologic picture is granular parietal IgG positivity, accompanied by C3 deposits in approximately 75% of cases. The IgG immunostaining usually is more intense than C3. The staining can be seen as large grains or densely grouped fine grains that gives a pseudolinear aspect. Observing in detail, it can be demonstrated that these deposits are located towards the external part of the GBM or “transmembrane”. Other immunoglobulins can also be identified in a minority of cases, specially IgM and IgA. As previously expressed, C1q or C4 deposits force to study a secondary cause of MGN; the same possibility must be considered if there are mesangial deposits.
IgG4 sub-class is the most frequent, this sub-class fixes poorly the complement and this would explain the weakest C3 staining (Doi T et al, Clin Exp Immunol 58:57-62, 1984 [PubMed link]; Nöel LH, et al, Clin Immunol Immunopathol 46:186-194, 1988 [PubMed link]).


Figure 10. MGN is characterized, immunopathologically, by IgG granular capillary wall deposits and, in most of cases, C3 in the same location. In thin sections is possible, in many cases, to determine the location of the immune deposits in the external part of the GBM. If we also find subendothelial deposits we must think about the possibility of a secondary MGN. In both images we can see a characteristic appearance of the subepithelial deposits, which we sometimes describe as "reticular aspect". (Immunofluorescence with antibodies anti-IgG marked with fluorescein, X400).
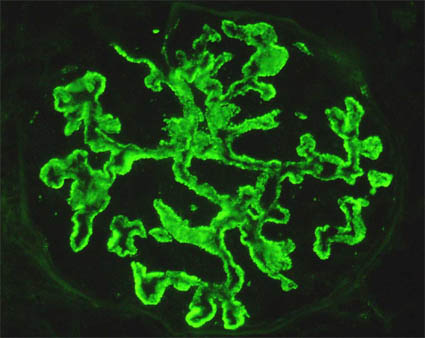

Figure 11. In some cases of MGN the parietal deposits are granules that seem to protrude towards the outside of the capillaries (as in the photo above). Other times we may find that the deposits are very small grains, densely grouped, that give a pseudolinear aspect (as in the bottom photo). At high power it is possible to determine the granular nature of the deposits and does not confuse them with linear deposits. (Immunofluorescence with antibodies anti-IgG marked with fluorescein, X400).
Electron microscopy
There are electron-dense deposits in the epithelial aspect (external) of the GBM, between this one and the epithelial cell: subepithelials or epimembranous. These deposits are usually diffuse and homogenously distributed, but they can be, in some cases, irregularly distributed. Spikes are demonstrated as irregular projections of the GBM among the subepithelial deposits; with progression of the disease these projections become longer and surround the deposits incorporating them in a thickened GBM. The deposits are amorphous; the presence of organized deposits must alert of a possible lupus nephritis. These deposits lose their electron density until disappear in the advanced stages of the process. Like in many other diseases with NS, there is a variable loss or effacement of podocyte foot processes. In some cases, more frequently secondary, there are dense deposits in mesangium. (Image of MGN stage II (link) - More EM images (link)

Figure 12a. Left: electron-dense deposits on the outside of the basement membrane, without reaction of this around the deposits: Stage I. Right: deposits surrounded laterally by material similar to that of the basement membrane ("spikes"): Stage II. Original magnification, X4,000. (Images courtesy of Dr. Carlos A, Jiménez).
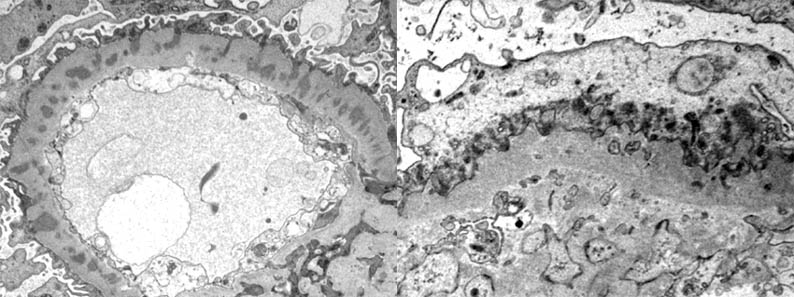
Figure 12b. Left: Electron-dense deposits are completely surrounded by basement membrane-like material, giving the appearance of being "embedded" within a very thick and irregular basement membrane: Stage III. Right: the glomerular basement membrane is thick and irregular and electron-dense deposits have almost disappeared: Stage IV. Original magnification: left, X4,000, right, X6,000. (Images courtesy of Dr. Carlos A, Jiménez). Note the extensive loss of pedicels in the four previous images.
Prognostic markers
Like in most glomerulopathies, serum creatinine
increase at diagnosis, severe proteinuria (>10 g/24h), arterial hypertension,
and chronic tubulointerstitial damage has been related, in greater or smaller
measurement, with a greater risk of evolution to terminal renal failure. Some
works suggest better prognosis if proteinuria is selective.
In secondary forms there is, in general, a better prognosis if the associate
cause has successful treatment. In children the prognosis seems better. In some
series there is better prognosis for women. Also the histopathologic stages
show correlation with the evolution, although this correlation is not perfect
as was previously expressed.
Recent bibliography
Homepage - - - Tutorial Index